Biosyngen received China NMPA IND approval for its T-cell redirection therapy targeting EBV-positive Lymphoma
April 7th, 2023, Biosyngen Pte Ltd (hereinafter as “Biosyngen”) was granted IND approval by China NMPA for the company’s first-in-class T-cell redirection therapy, it is an autologous T cell therapy for EBV-positive lymphoma. The principle of autologous T cell therapy is to genetically modify patients' own T cells to express additional receptors for Epstein-Barr virus (EBV) antigen recognition and T cell activation upon EBV+ tumor cell engagement, targeting cancer indications.
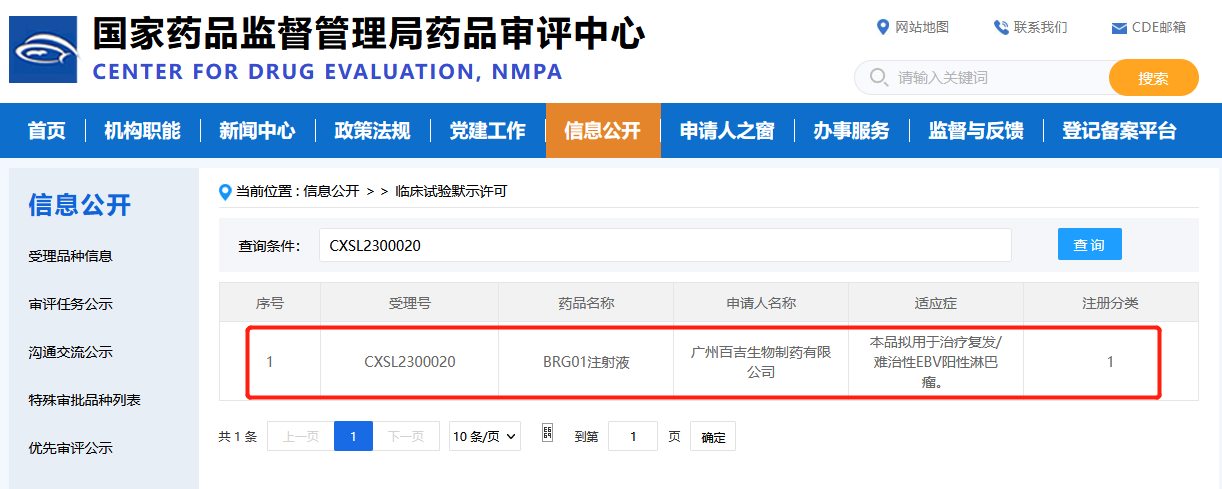
Related:https://www.cde.org.cn/main/xxgk/listpage/4b5255eb0a84820cef4ca3e8b6bbe20c
Currently, commercially available cell therapies, such as CD19 CAR-T, are designed for the treatment of B cell lymphoma or acute lymphoblastic leukemia. Treatment options for EBV-associated lymphoproliferative diseases and lymphoma are limited and in urgent demand. The company’s new pipeline targeting EBV-associated lymphoma is a hope for this population of patients. “Biosyngen has been committing itself to high quality R&D of cell therapy. This is the second IND approval obtained for the company’s first-in-class products worldwide, demonstrating our commitment to remain focused on unmet clinical needs of cancer patients,” said Dr. Michelle Chen, COO of Biosyngen. “At this point, in addition to this announcement, we have secured IND approvals for nasopharyngeal carcinoma treatment by US FDA and China NMPA. Following closely will be news of Biosyngen’s application to FDA for the treatment of EBV positive lymphoma. In addition, preparation for applications of orphan drug and Fast Track eligibility are also well under way.” Biosyngen has continued in building up a pipeline of other cancer indications; within 2023, the company made plans for IND applications for other therapies such as lung cancer and liver cancer across key regions – Singapore, the US and China.
About EBV-positive Lymphoma
EBV, the first oncovirus identified, is a human herpesvirus and has infected ~95% of global population. It has been listed as Group 1 carcinogen ("Carcinogenic to humans") by World Health Organization (WHO) and proved to be associated with a range of diseases including nasopharyngeal cancer, EBV-positive gastric cancers, lymphoma and lymphoproliferative diseases. EBV is so markedly lymphotropic that, for those who suffer from impaired immune system, especially T cell function, EBV may even induce lymphoblastic malignancies. EBV reactivation was also observed in patients who received bone marrow transplantation and CD7 CAR-T cell therapy, who may even develop EBV positive lymphoma.
The therapy developed by Biosyngen is an engineered T cell therapy, also known as a type of adoptive immune cell therapy indicated for nasopharyngeal cancer and EBV-positive lymphoma. Patients' T cells are isolated and genetically modified in a GMP-compliant facility to enhance their ability to recognize and attack specific antigens on cancer cells. The modified T cells are expanded ex vivo and infused back into the patient. The infused T cells would bind to specific antigen on the cancer cells to mediate tumor killing. The preliminary safety and efficacy of BRG01 Therapy have been demonstrated in data from exploratory clinical trials.
The scientific direction of Biosyngen is focused on targeting multiple solid tumors and hematological tumors. The company has independently developed a number of exclusive technical platforms specifically for cancer immunotherapy, including IDENTIFIER, SUPER-T and MSE-T. These platforms are designed for improved safety and efficacy, equipping the company with capabilities to overcome challenges in antigen identification, antibody TCR screening and identification of immune cell function


